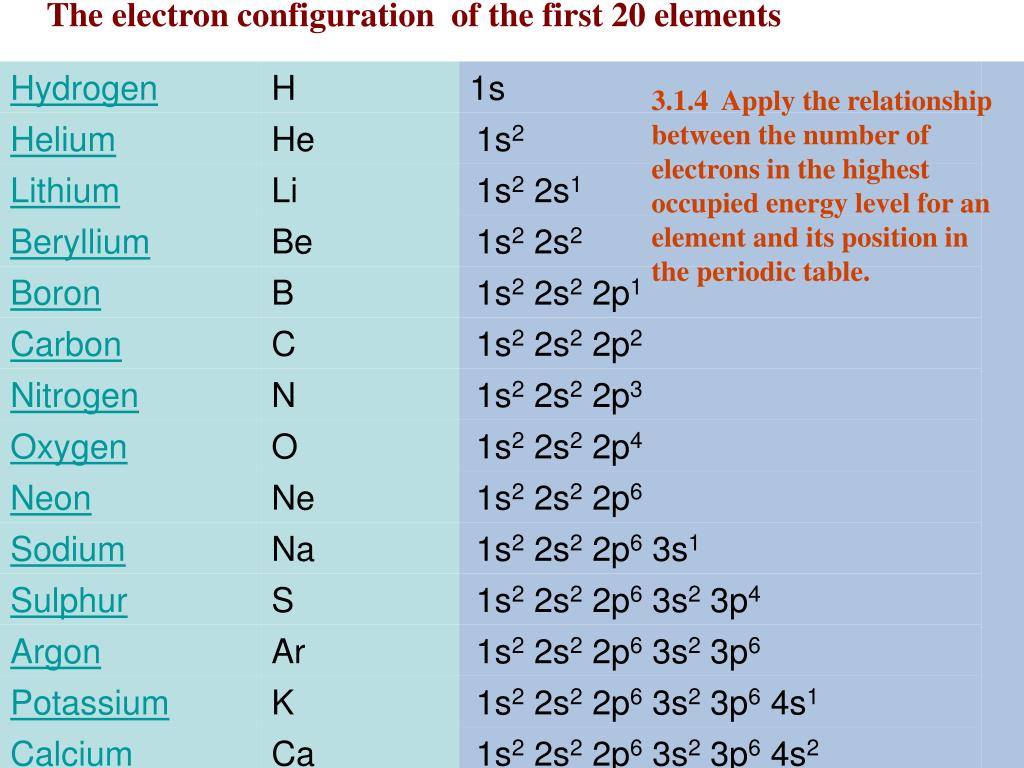
You can effortlessly find every single detail about the elements from this single Interactive Periodic table. The positively charged nucleus is surrounded by a cloud of negatively charged electrons. Let me tell you how this Interactive Periodic Table will help you in your studies.ġ). About 50 years after Mendeleevs first periodic table, scientists discovered the atom was built around a nucleus with positively charged protons and neutral neutrons - both of which are relatively heavy. Free Gift for you: Interactive Periodic Table In this way, the elements of the same group show similar chemical properties and they also have the same number of valence electrons. They are soft and can be cut easily with a kitchen knife.Īlso all the elements of group 1 have one valence electron.Īll the elements of group 18 are chemically inert (that means they do not easily react with other elements).Īnd all the elements of group 18 have a complete octet (that means they have 8 electrons in their outer shell). The elements lying in the same groups show similar chemical properties and they also have same number of valence electrons.Īll the elements of group 1 are highly reactive to water. There are total 18 vertical columns on periodic table. Copper is also needed for several proteins to function properly in the body.Groups are the vertical columns on the periodic table. The number of valence electrons depends on the octet rule. (Recall that the number of valence electrons is indicated by the position of the element in the periodic table. As a result, elements in the same group often display similar properties and reactivity. The elements in each group have the same number of valence electrons. Add together the valence electrons from each atom. The s-, p-, and d-block elements of the periodic table are arranged into 18 numbered columns, or groups. Elements within the same group share the same number of valence electrons. Determine the total number of valence electrons in the molecule or ion. Atoms in a period have the same number of electron shells. Zinc is needed for the body’s immune system to function properly, as well as for protein synthesis and tissue and cell growth. An element period is a horizontal row on the periodic table. We can write the configuration of oxygen's valence electrons as 2s2p. For example, oxygen has six valence electrons, two in the 2s subshell and four in the 2p subshell. Other transition metals have important functions in the body, despite being present in low amounts. About Transcript Valence electrons are the electrons in the outermost shell, or energy level, of an atom. It is the presence of this particular transition metal in your red blood cells that allows you to use the oxygen you inhale. The increasing positive charge attracts the electrons more strongly, pulling them closer to the nucleus. Each hemoglobin molecule has four iron atoms, which act as binding sites for oxygen. As we go across a period on the periodic table, however, electrons are being added to the same valence shell meanwhile, more protons are being added to the nucleus, so the positive charge of the nucleus is increasing. The crucial atom in the hemoglobin protein is iron. Hemoglobin is a relatively large molecule, with a mass of about 65,000 u. Hemoglobin combines with oxygen and carbon dioxide, transporting these gases from one location to another in the body. The critical part of the red blood cell is a protein called hemoglobin. 
Without red blood cells, animal respiration as we know it would not exist. Red blood cells are cells that transport oxygen from the lungs to cells of the body and then transport carbon dioxide from the cells to the lungs. The chemistry of iron makes it a key component in the proper functioning of red blood cells. Transition metals have interesting chemical properties, partially because some of their electrons are in d subshells. Because iron has relatively massive atoms, it would appear even lower on a list organized in terms of percent by atoms rather than percent by mass. In the second period elements, the two electrons in the 1 s sublevel are called inner-shell electrons and are not involved directly in the element's reactivity, or in the formation of compounds. 
The first element appearing on the list that is not a main group element is iron, at 0.006 percentage by mass. Valence electrons are the electrons in the highest occupied principal energy level of an atom. Most of the elemental composition of the human body consists of main group elements. To Your Health: Transition Metals in the Body Each group is located in a different part of the periodic table. Elements are either metals, nonmetals, or semimetals.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
